Electrocatalyst for hydrogen production
Electrocatalyst for hydrogen production for greener solutions
Various researches on electrocatalyst for hydrogen production
Most-efficient, longer-lasting and lightweight batteries for electric vehicles is a major concern all over the world. From smartphones to electric vehicles the demand for lithium-ion batteries is in high demand. As lithium is a rarely found metal for many countries like India, it has become a hurdle for manufacturing batteries. But there are many alternatives to lithium like hydrogen, we can work to excel the technology to produce at large scale and at low cost. Let us see the possibilities of hydrogen in the form of fuel cell vehicles.
Electrocatalyst for hydrogen production
There have been many kinds of research done in past and in present, there are a lot of researchers working on hydrogen to make use of hydrogen as it is a clean energy source for the future. Finding an alternative way for fossil fuels and other natural gases which are very harmful, the alternate solution becomes necessary to reduce the pollution levels from the country.
IIT Madras collaborated with German Researchers to develop new materials-Project SPARC
IIT Madras researchers have collaborated with German Researchers to develop new materials for Green Energy Solutions. The project aims to develop alternative technologies to produce Green Hydrogen. The project has been taken under the Scheme for Promotion of Academic and Research Collaboration (SPARC) which is an initiative by the Ministry of Human Resource Development, Government of India. SPARC project holds a budget of 66 lakh rupees by the government.
A team of researchers led by Prof. N.V. Ravi Kumar from the Department of Metallurgical and Materials Engineering at IIT Madras with the collaboration Prof. Sanjay Mathur, Chair of Inorganic and Materials Chemistry, Institute of Inorganic Chemistry, University of Cologne, Germany and other researchers.
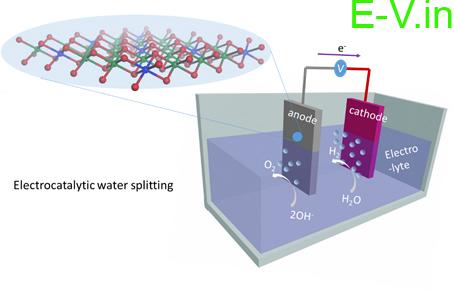
The team head Prof. N.V. Ravi Kumar said that “Conventional methods of generating hydrogen (H2) such as ‘water-gas conversion’ and ‘partial oxidation of hydrocarbons’ generate a large quantity of carbon dioxide (CO2), a notorious greenhouse gas that imposes serious environmental concerns. In contrast, electrochemical splitting of water termed as ‘Water Electrolysis’ (WE) is a clean, facile, and highly efficient technology for large-scale production of high-purity H2. Through this international collaboration, we aim to develop novel low-cost electrocatalysts for hydrogen evolution reactions.”
The team is working together to understand the advanced electrocatalysts with the potential to transform lab-scale research to deployable reactors/devices.
Their research result for this project is that the In decarbonisation of energy technologies Hydrogen based energy transition is a central pillar. Present hydrogen production technologies need new materials and hydrogen-production pathways involving earth-abundant resources such as water, solar energy, and light-absorbing pigments.
The SPARC project’s aim is to develop a holistic approach for identifying next-generation photo-electrocatalysts by considering inputs from first principle calculations and through phase diagram calculations which is a unique and unexplored approach in the field of photo-electrocatalysis said Prof.Sanjay Mathur (one of the Adjunct Faculty Members of the Department of Metallurgical and Materials Engineering, IIT Madras).
Indian scientists develop bio-inspired electrocatalyst to develop efficient, low-cost energy conversion & storage technologies
According to PV magazine India, Indian scientists have synthesised a bio-inspired electrocatalyst to develop efficient, low-cost energy conversion and storage technologies such as fuel cells, biofuel cells, and metal-air batteries. The electrocatalyst is developed by Dr Ramendra Sundar Dey and his team at the Institute of Nano Science and Technology (INST), Mohali which is based on iron, manganese and N-doped fish gill derived carbon. It has a unique porous structure and could provide conductive carbon networks after heat treatment.
The catalyst was used as an air cathode in the zinc-air battery, was able to show active oxygen reduction reaction (ORR) in a wide range of pH (pH< 1, 7, and >13) and outperformed platinum-on-carbon (Pt/C) catalyst.
They have found a fabricated a homemade rechargeable Zn-air battery (ZAB) with the catalyst as an air cathode. It showed an almost stable charge and discharge voltage plateaus after rigorous cycling for a long duration. It surpassed the commercial Pt/C based ZAB performance.
The reason behind the amazing performance of the catalyst is the presence of the Fe-Mn based binary moiety which benefits for the oxygen binding and boosting ORR catalytic performances in alkaline medium by weakening the Oxygen-Oxygen bonds.
low-cost, high-performance multinary intermetallic compound as an active electrocatalyst for hydrogen production
In another case, a team of scientists from City University of Hong Kong (CityU) co-led by Professor Lu Jian, CityU’s Vice President (Research and Technology) and Chair Professor of Mechanical Engineering, and Professor Liu Chain-tsuan, University Distinguished Professor of the College of Engineering.
The electrochemical water splitting is a relatively environmentally friendly process when compared to the other methods of hydrogen production. At present most of the developed electrocatalysts are noble-metal based, such as platinum and palladium which is costly and rarely found which is hindering the development and applications of this hydrogen production method.
Professor Liu’s team partnered with Prof Lu’s team, experts in noble metal research, and created a new high-entropy intermetallic (HEI) electrocatalyst by adopting the aforementioned alloy design strategy. The new electrocatalyst mainly comprises iron, cobalt, nickel, aluminium and titanium.
A simple one-step chemical method has been used and the team produced a dendrite-like porous structure which greatly increased the surface area for electrochemical activities and hence significantly enhancing the electrochemical performance. Because of its unique constituents and structure, this HEI catalyst performs excellent hydrogen evolution reaction in alkaline electrolyte.
When hydrogen is consumed in a fuel cell it produces only water without generating carbon dioxide. Therefore it is said to be a clean source to reduce the amount of pollution in the air. The production of hydrogen by electrochemical water splitting is more environmentally friendly when compared to other methods. However, at current the electrocatalysts are noble-metal based, such as platinum and palladium which are very expensive and scarcity hinders the development and application of this hydrogen production method. Hopefully, we will be able to find some great solutions and alternates to produce hydrogen so that it can be used in as many as vehicles which results in reducing the pollution levels in the country.
If you are an EV manufacturer or EV Dealer or EV Supporter who want to share news related to electric vehicles on our website, please send an email to crm@electricvehicles.in
For the latest electric vehicles news, follow electricvehicles.in on Twitter, Instagram,Facebook and subscribe to our YouTube Channels English, Hindi, Telugu and Kannada




